You separate the two with something that acts neutral between the two parties, called a separator. On each side of the paper, you put the material you want to use as a cathode and anode. You would be surprised about the amazingly large surface area you have in a small household battery if you unrolled it. In a normal cylindrical household battery that you have at home for the remote control, for example, that large surface is created with a thin, long, paper-like material that is rolled up and put in a can. Think about it like a short or a long river, with more or less water. To create a lot of current and to store a lot of energy with electrons requires a lot of surface area where the anode and cathode face each other. Many materials, mostly metal we use on Earth, are suitable for it, and one of the best is lithium, which is nothing other than an ordinary metal that you find almost everywhere on earth. To find a medium that creates a steady and reliable flow of electrons can be called finding good cell chemistry. 
You just learned what Volt and Ampere are, two other members of our Greek drama. If the energy is the amount of water and your river is narrow, it needs to flow with higher speed or current to have the same energy/water supplied as if it’s a wide river but the current is flowing slowly. To make use of it for an electronic device, you need to choose a medium that is doing a good job at holding a lot of electrons and at making them flow to the other side, which creates what is called a current, like a river current that flows steadily in one direction. What sounds like names out of a Greek drama is as simple as that. Because having a few hundred electric eels in a tank in your car to power it is not a working plan, a different medium was searched for to hold electrons, and that was metal.Īn anode and cathode are nothing other than a place where we have more or fewer electrons in a medium. The famous inventor Nikola Tesla wanted to make nature a part of the electricity supply, and although he proved it works, he had too many enemies and no commercial model to scale it. Nature is diverse and some animals make use of electricity like for instance the electric eel but there are many more. The reason why we call a battery a battery is simply because we designed them to be able to store more electrons than we usually find in nature to make use of it. Whoever has had their hair stand up or got an electric hit from a cow fence or another person knows how it feels when electrons suddenly flow – it can hurt.īatteries surround us and we are sometimes batteries too, therefore, batteries are everywhere and normal. Even we humans sometimes behave like a battery. 
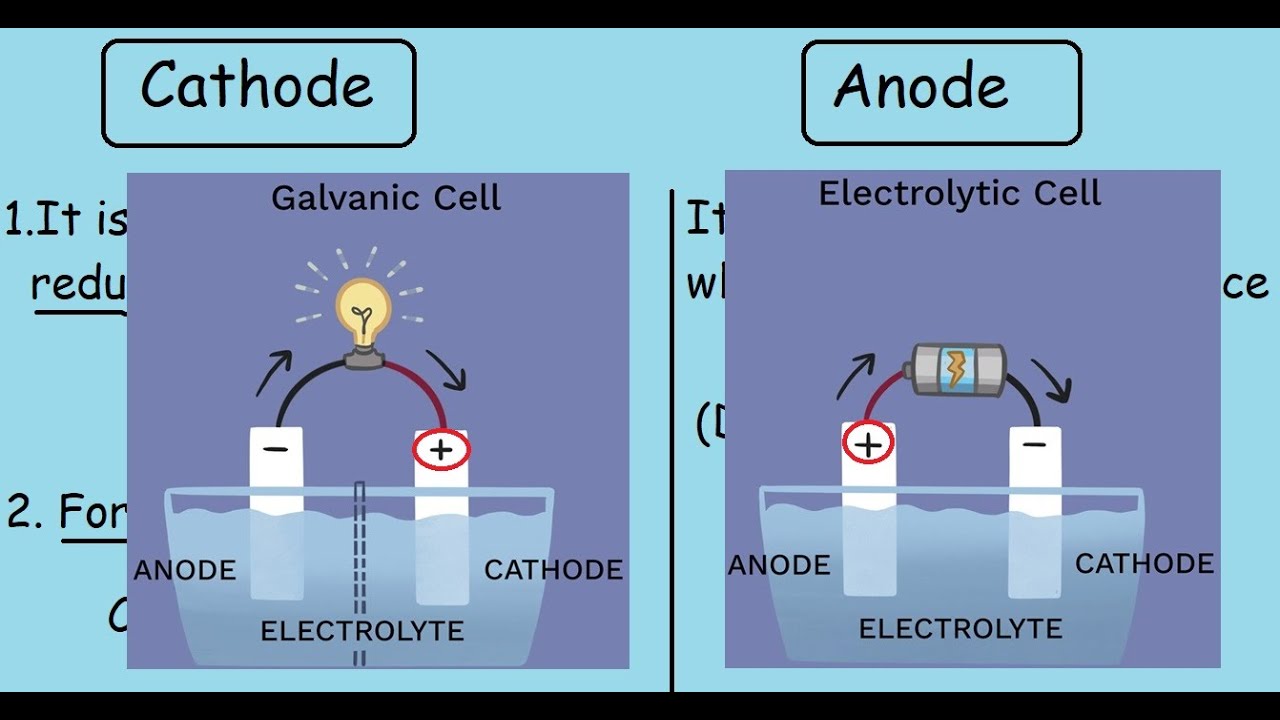
The anode and cathode are just the two names that describe the place where the electrons either move to or come from.Īll and everything in our world has electrons, and all and everything has at a certain point in time more electrons or fewer electrons, behaving like a battery. Forget all sophisticated technical terminology and measures, because that is all you need to know. You either make energy available for whatever electric device you use it for, or you put energy in the medium to store it for later use. While you do that, you charge or discharge a battery with energy. What the heck is an anode and cathode in a battery, and why is that important?Ī battery is nothing other than a medium in which you move electrons from one side to the other.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
